Summary
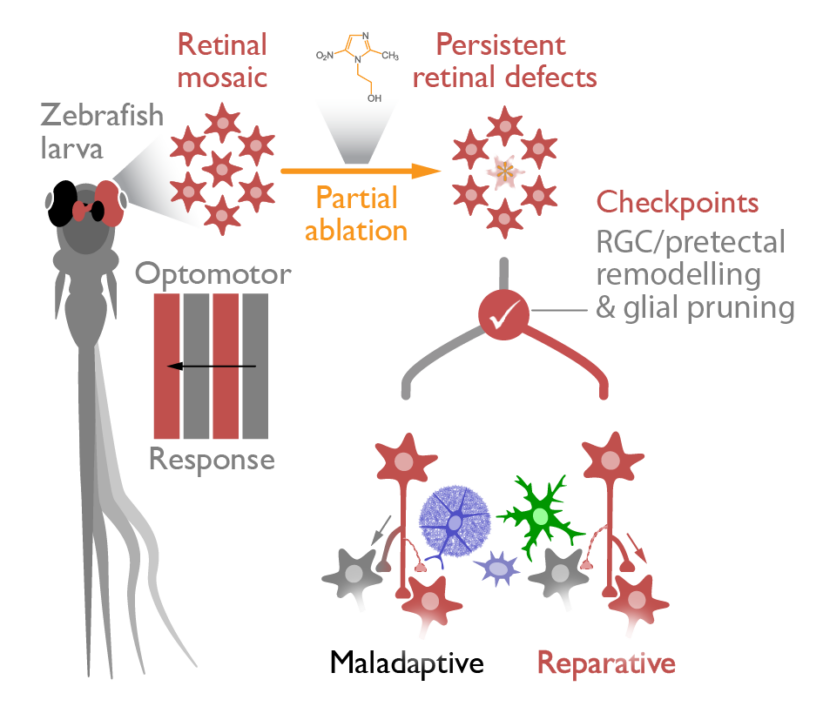
Circuit remodelling following brain trauma represents compensatory changes that may restore lost function. We investigate network remodelling in the visual system of zebrafish as it allows an unbiased systems-based approach to identify the changes and checkpoints that enable compensatory circuit repair. After ablating specific retinal photoreceptors, we will probe changes in network activity and structural remodelling in central visual circuits, as well as investigate the contribution of glial cells to synaptic plasticity to reveal the mechanisms mediating the recovery of visual function.





