Background: Microglia are key regulators of tissue plasticity and functional recovery in the chronic phase after ischemic stroke.
Hypothesis: We hypothesize that microglia are a central hub in the multi-cellular recovery process after stroke; thereby, microglia represent a checkpoint to modulate tissue homeostasis based on complex multi-cellular interactions.
Strategy: We will perform a multi-omic characterization of the cellular network during stroke recovery and focus on the role of microglial interactions with immune and glial cells which have pro-regenerative functions.
Summary
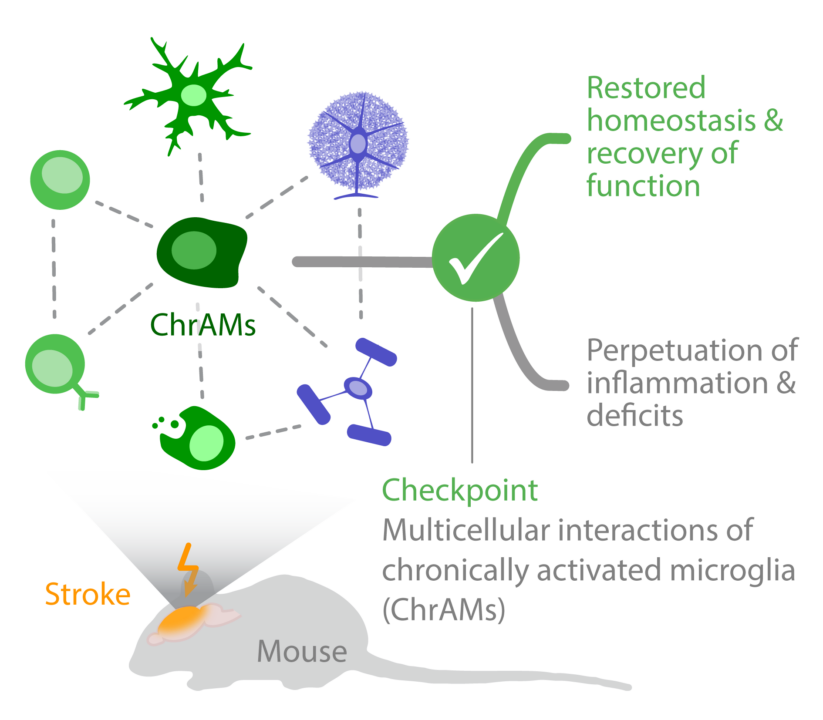
Ischemic brain lesions not only induce an acute inflammatory response to the tissue injury but result in chronic neuroinflammation which is insufficiently resolved. Post-stroke neuroinflammation is largely detrimental in the acute phase after stroke and contributes to secondary lesion expansion. Immune cell populations and inflammatory mechanisms of the acute response to stroke has been well characterized in previous work. However, the contribution of chronic post-stroke neuroinflammation to functional recovery and the underlying mechanisms of insufficient resolution of inflammation are barely investigated. In the previous funding period, we identified a subpopulation of chronically activated microglia (ChrAMs) after stroke, which are distinct from acutely reactive as well as other previously described microglia identities. Surprisingly, depletion of microglia in the chronic phase after stroke substantially impaired functional recovery, but the specific modulation of microglial activity state by non-depleting approaches was able to improve long-term outcome. Microglia depletion or modulation affected not only neuronal function but also multiple other cell populations as we observed in our preliminary experiments changes in myelination, synapse pruning and leukocyte recruitment. Based on these preliminary findings, we propose that microglia are cellular checkpoints of tissue homeostasis and plasticity in the chronic post-stroke phase by interaction with multiple other cell populations. As a result of this central microglial function, its chronic reactivity can result in a self-perpetuating process of chronic neuroinflammation and impaired tissue restoration, while modulating microglial activity can support the restoration of tissue homeostasis. Hence, the goal of this project is to identify the cellular interactions of microglia during post-stroke recovery by defining the cellular interaction partners, mechanisms of interaction and the impact of microglia on multicellular network function. To identify these multicellular interactions, we will utilize genetic animal models and pharmacological approaches to modulate cell-cell interactions as well as human brain tissue samples for translational validation. We will perform multiomic analysis including spatial transcriptomics of immunological synapses, proteomics of the microglial secretome and metabolome analysis. Post-stroke recovery will be studied by combination of behaviour tests and in vivo imaging of cortical network function.





