Summary
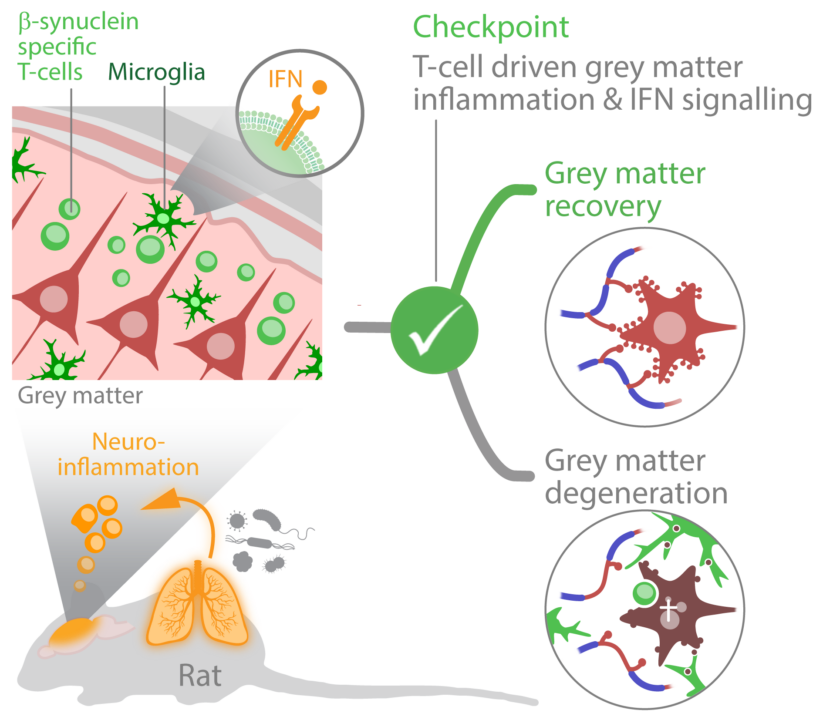
This project focuses on recovery mechanisms of the CNS tissue in the course of acute T-cell mediated autoimmune inflammation. We hypothesize that T-cell induced changes of the grey matter are crucial for the failing recovery of the CNS after repeated autoimmune bouts and that lung microbiota are external regulators of de- and regenerative processes within the CNS tissue. We aim to identify brain-resident and peripheral targets to prevent permanent CNS damage and/or to foster its recovery which might in perspective be exploited to develop novel therapeutic strategies.