Summary
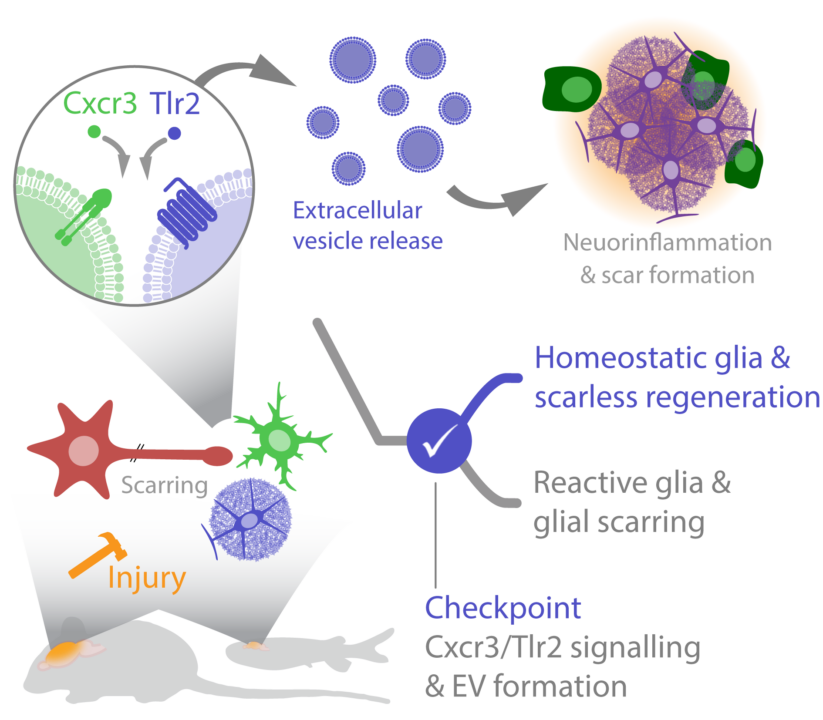
Traumatic brain injuries (TBIs) cause primary tissue damage and activate glial cells to build glial border, thereby delineating the injured brain parenchyma from the healthy surrounding tissue. However, this necessary primary reaction of glial cells often persists. This long-lasting glial activation results in prolonged and exacerbated neuroinflammation that prevents tissue restoration. We propose that a cross-regulatory network between innate immunity pathways and EV cargo defines an important checkpoint that turns protective glial border cells into tissue scaring cells. We aim to identify EV cargos and their dependence on innate immunity pathways as drug-targetable molecular checkpoints to improve tissue regeneration.