Summary
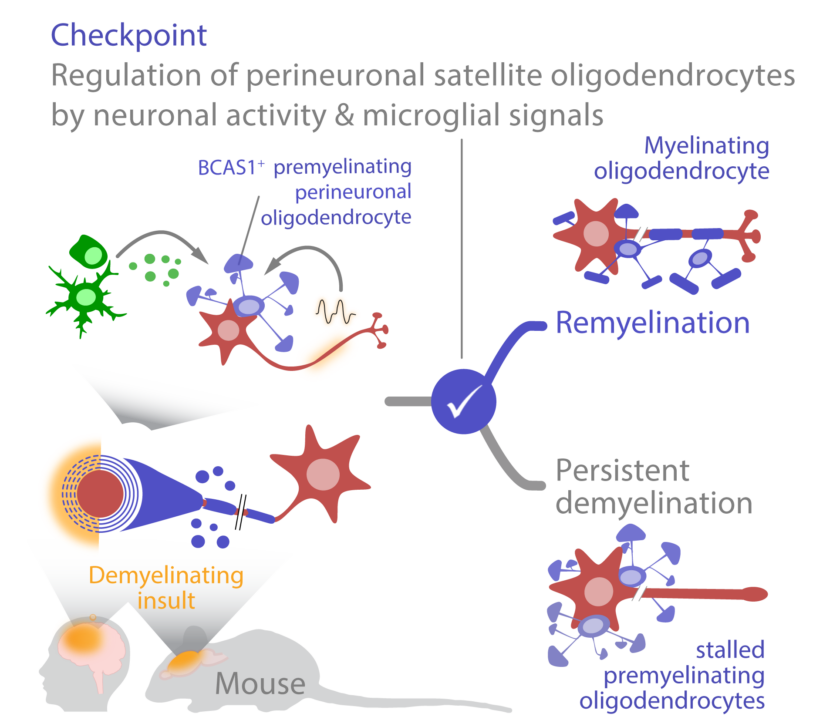
The cerebral cortex contains a special, perineuronally localized population of oligodendrocyte lineage cells with possible functions in connectivity and myelination. We hypothesize that this cell population serves as a reservoir of remyelinating cells after myelin damage. We will apply spatial transcriptome analysis with subcellular resolution in experimental models of demyelination and human tissue from patients with multiple sclerosis to define physiological and pathological states of perineuronal oligodendrocytes. In addition, we will study checkpoints of satellite cell recruitment in experimental models by lineage tracing, in vivo imaging, and chemogenetic modulation of neuronal activity.





