Summary
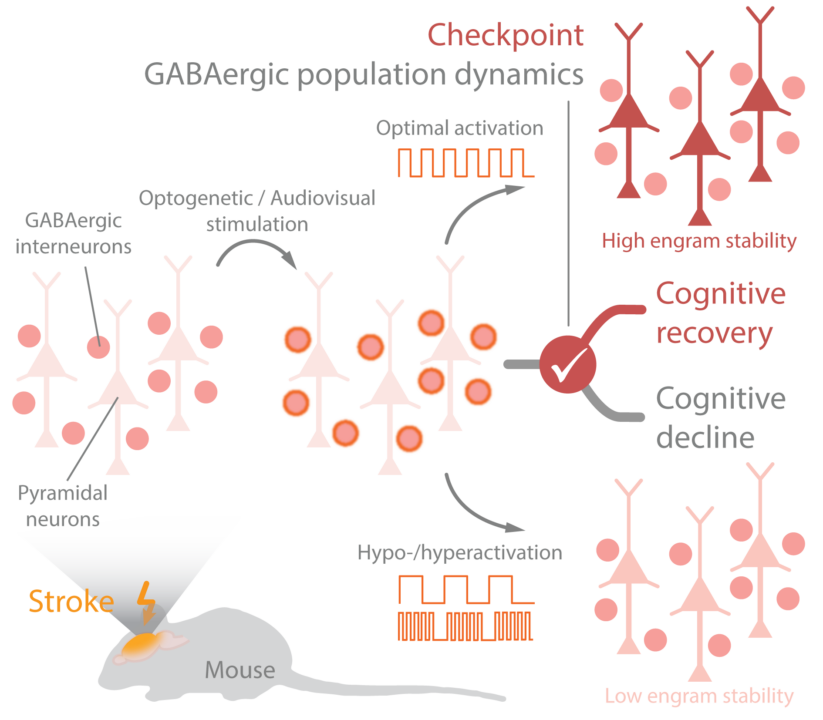
Strokes destroy excitatory inputs – not only in the stroke area, but also in remote regions such as the hippocampus, which may lead to disorganized GABAergic activity and cause cognitive decline. We propose here to identify and modulate checkpoints of GABAergic population dynamics shaping microcircuits of excitatory pyramidal neurons in the hippocampus. We aim at enhancing functional and structural rewiring processes by systematically modulating GABAergic interneuron activity and thus defining novel therapeutic targets for preventing cognitive decline after stroke.





