Summary
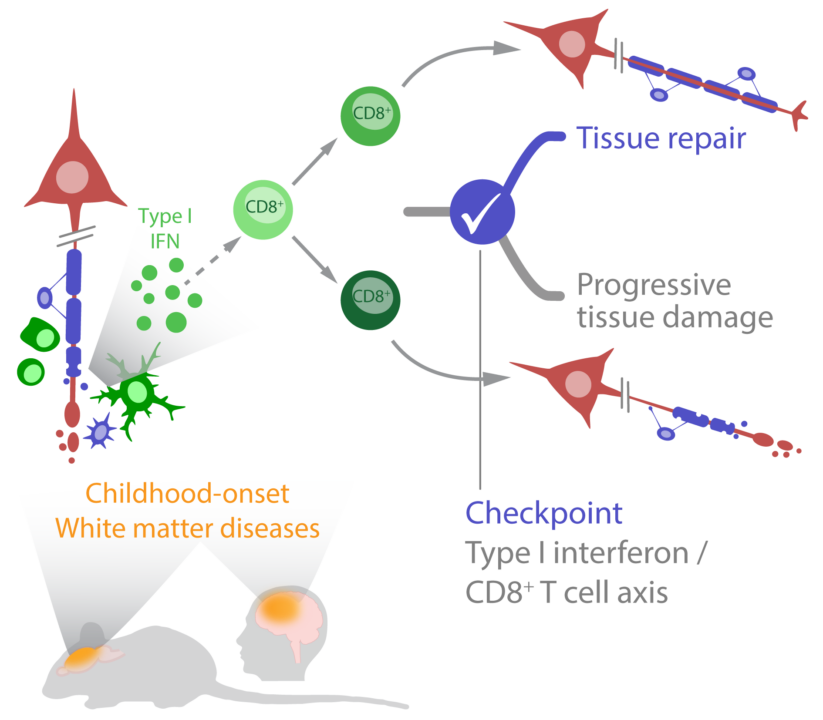
Childhood-onset white matter diseases including multiple sclerosis and monogenetic leukoencephalopathies, are characterized by neuroinflammatory tissue damage. Here, we propose that CD8+ T cell phenotypes within lesions serve as critical immune checkpoints for tissue damage and regeneration. Our objective is to define CD8+ T cell-driven disease and regenerative pathways by utilizing a combination of multifluorescent immunohistochemistry, spatial transcriptomics and multimodal MR imaging in patients and preclinical models. We expect to identify common CD8+ T cell driven inflammatory mechanisms for these diseases and thus to pave the way for novel therapeutic approaches.